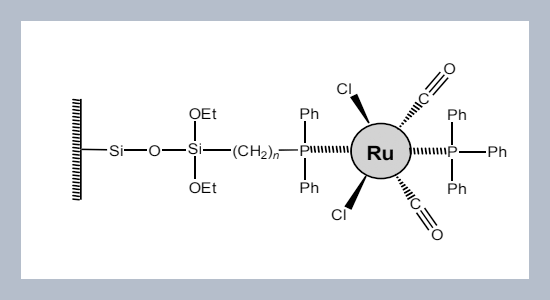
REFERENCES
- [1] American Oil Chemists’ Society. 1988. “Official Methods and Recommended Practices of the American Oil Chemists’ Society”. 3rd Ed., AOCS Press. Champaign, L.
- [2] Asherio, A., Katan, M. B., Zock, P. L., Stampfer, M. J., and Willett, W. C. 1999. Trans fatty acids and coronary heart disease. New England Journal of Medicine, 340: 1994-1998.
- [3] Azran, J., Buchman, O., Orchin, M., and Blum, J. 1984. Polymer-bound iridium catalysis. Hydrogen transfer from formic acid to unsaturated carbon-carbon bonds. Journal of Organometallic Chemistry, 49: 1327-1333.
- [4] Bello, C., Diosady, L. L., Graydon, W. F., and Rubin, L. J. 1985. Homogeneous catalytic hydrogenation of canola oil using a ruthenium catalyst. Journal of the American Oil Chemical Society, 62: 1587-1592.
- [5] Bailar, J. C. Jr. 1975. Heterogenizing homogeneous catalysts. Catalysis Reviews-Science and Engineering, 10: 17-36.
- [6] Capka, M. 1977. Catalysis by Metal Complexes. XLV. Selective phosphination of (2-chloropropyl) triethoxysilane, a new route to alkoxysilyl-substituted phosphines. Synthesis and Reactivity in Inorganic and Metal-organic Chemistry, 7: 347.
- [7] Czakova, M. and Capka, M. 1981. Hydogenation Activity of homogeneous and heterogenized rhodium (I) complexes containing {w-(triethoxysilyl) alkyl} aliphenylphosphines. Journal of Molecular Catalysis, 11: 313.
- [8] Evans, G. O., Pittman, C. U., McMillan, R., Beach, R. T., and Jones, R. 1974. Synthetic and catalytic studies of polymer-bound metal carbonyls. Journal of Organometallic Chemistry, 67: 295.
- [9] Grubbs, R. H., Kroll, L. C., LeRoy, C., and Sweet, E. M. 1973. Preparation and selectivity of a polymer-attached rhodium (I) olefin hydrogenation catalyst. Journal of Macromolecular Science-pure and Applied Chemistry, 7: 1047.
- [10] Hu, F. B., Stampfer, M. J., Manson, J. E., Rimm, E., Colditz, G. A., Rosner, B. A ., Hennekens, C. H., and Willett,W. C. 1997. Dietary fat intake and the risk of coronary heart disease in women. New England Journal of Medicine, 337: 1491-1499.
- [11] Mensik, R. P. and Katan, M. B. 1990. Effect of dietary trans fatty acids on High-density and Low-density lipoprotein cholesterol levels in healthy subjects. New England Journal of Medicine, 323: 439-445.
- [12] Michalska, Z. M., Capka, M., and Stoch, J. 1981. Silica-supported rhodium complexes. relation between catalyst structure and activity. Journal of Molecular Catalysis, 11: 321-330.
- [13] Pittman, C. U. and Evans, G. O. 1973. Polymer-bound catalysts and reagents. Chemiscal Technology: 560.
- [14] Pittman, C. U., Smith, L. R., and Hanes, R. M. 1975. Catalytic reactions using polymer-bound versus homogeneous complexes of nickel, rhodium, and ruthenium. Journal of the American Oil Chemical Society, 97: 1742-1748.
- [15] Sanchez-Delgado, R. A., Duran, , Monfort, J., and Rodriquez, E. 1981. A comparative study of the isomerization and hydrogenation of 1-hexene with soluble and silica-supported ruthenium catalysts. Journal of Molecular Catalysis, 11: 193.
- [16] Skupinski, W. and Malinowski, S. 1975. Catalytic systems containing nickel supported on silica and bonded with organic ligands and complexed by Lewis acids. Journal of Organometallic Chemistry, 99: 465.
- [17] Skupinski, W. and Malinowski, S. 1976. Investigations on catalytic systems containing nickel supported on silica gel and bonded with organic ligands and complexed by Lewis acids (II). Journal of Organometallic Chemistry: 117-183.
- [18] Strukul, G., Bonivento, M., Graziani, M., Cernia, E., and Palladino, N. 1975. Selective hydrogenation of ketones and olefins over polymer-bound rhodium catalysts. Inorganica Chimica Acta, 12: 15-21.
- [19] Williamson, K. L. 2003a. “Macroscale and Microscale Organic Experiments. Chapter 38: Grignard Synthesis of triphenylmethanol and benzoic acid”. 4th Ed., Houghton Mifflin Company, Boston, Massachusetts: 461-478.
- [20] Williamson, K. L. 2003b. “Macroscale and Microscale Organic Experiments. Chapter 24: Oxidation with pyridinium chlorochromate”. 4th Ed., Houghton Mifflin Company, Boston, Massachusets: 325-334.
- [21] Wright, A. J., Mihele, A. L., and Diosady, L. L. 2003. Cis selectivity of mixed catalyst systems in canola oil hydrogenation. Food Research International, 36: 797-804.