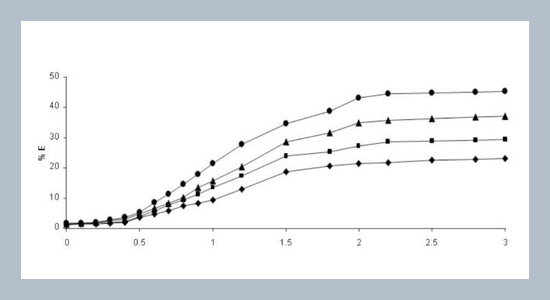
REFERENCES
- [1] Colonia, E. J., Dixit, A. B., and Tavare, N. S. 1998. Separation of chlorobenzoic acids through hydrotropy. Industrial and Engineering Chemistry Research, 37: 1956-1969.
- [2] Badwan, A.-A. El-Khordagui, L.-K., and Saleh, A.-M. 1983. The solubility of benzodiazepines in sodium salicylate solutions and a proposed mechanism for hydrotropic solubilization. International Journal of Pharmaceutions, 13: 67-74.
- [3] Saleh, A.-M. and El-Khordaugi, L.-K. 1985. Hydrotropic agents. International Journal of Pharmaceutions, 24: 231-238.
- [4] Balasubramanian, D., Srinivas,V., Gaikar, V.-G., and Sharma, M. -M. 1989. Aggregation behavior of hydrotropes in aqueous solutions. Journal Physical Chemistry, 93: 3865-3870.
- [5] Agarwal, M. and Gaikar, V.-G. 1992. Extractive separation using hydrotropes. Separations Technology, 2: 79-84.
- [6] Liaonanchen, X. and Micheau, J.-C. 2002. Hydrotrope induced autocatalysis in the biphasic alkaline hydrolysis of aromatic esters. Journal of Colloid and Interface Science, 249: 172-179.
- [7] Marcoz Spitzer, H. and Desilva, R. C. 1999. Investigations on the mechanism of aqueous solubility increase caused by some hydrotropes. Journal of Thermochemical, 28: 161- 167.
- [8] Gnanendran, N. and. Amin, R. 2002. The Effect of hydrotropes on gas hydrate formation. Journal of Petroleum Science and Engineering, 40: 37-46.
- [9] Gaikar, V.-G. and Sharma, M.-M. 1986. Extractive separation with hydrotropes. Solvent Extraction and Ion Exchage, 4: 839-846.
- [10] Gaikar, V.-G. and Sharma, M.-M. 1993. Separation with hydrotropes. Separations Technology, 3: 2-11.
- [11] Korparkar, Y. -P. and Gaikar, V.-G. 2004. Solubility of o-/p- hydroxyacetophenones in aqueous solutions of sodium alkyl benzene sulfonate hydrotropes. Journal of Chemical and Engineering Data, 49: 800-803.
- [12] Gandhi, N. N. and Kumar, M. D. 1998. Solubility and mass transfer coefficient enhancement of ethyl benzoate through hydrotropy. Hung- arian Journal of Industrial Chemistry, l, 26: 63-68.
- [13] Roy, B.-K. and Moulik, S.-P. 2003. Effect of hydrotropes on solution behavior of amphiphiles. Journal of Current Science, 85: 23-28.
- [14] Taboada, B. P. Castro, E., and Mosquera, V. 2006. Influence of SDS and two nonionic hydrotropes on the micellized state of the triblock Copolymer. Journal of Colloid and Interface Science, 296: 677–684.
- [15] Hodgdon, T. K. and Kaler, E. W. 2007. Hydrotropic solutions. Journal of Colloid and Interface Science, 12: 121-128.
- [16] Gandhi, N. N. and Meyyappan, N. 2005. Solubility and mass transfer coefficient enhancement of benzyl benzoate in water through hydrotropy. Journal of Chemical and Engineering Data, 50: 796-800. Friberg, S. E., Fei. L., Campbell, S., and Lu, Y. 1997. Vapor pressure of phenethyl alcohol in an aqueous hydrotrope solution. Journal of Colloid and Interface Science, 127: 233-239.
- [17] Rigoli, I. C., Schmitt, C. C., and Miguel, G. 2007. The hydrotrope effect on the photo- polymerization of styrene sulfonate initiated by Ru complexes. Journal Photochemistry and Photobiology Chemistry, 188: 329-333.
- [18] Gandhi, N. N. and Meyyappan, N. 1998. Effect of Hydrotropes on solubility and mass- transfer coefficient of butyl acetate. Journal of Chemical and Engineering Data, 43: 695-699.
- [19] Colonia, E. J. and Tavare, N. S. 1997. Separation of eutectics of chloronitrobenzenes through hydrotropy. Journal of Chemical and Engineering Data, 42: 631-635.
- [20] Bauduin, P., Renonncourt, A., Kopf, A., and Kunz, W. 2005. Unified concept of solubilization in water by hydrotropes and cosolvents. Langmuir, 21: 6769-6775.
- [21] Gandhi, N. N., Ramesh. N., and JayaKumar, C. 2009. Effective Separation of petroproducts Through Hydrotropy. Journal of Chemical Engineering Technololgy, 32: 1-6.
- [22] Gaikar, V.-G. and Phatak, P.-V. 1999. Selective solubilization of isomers in hydrotrope solutions: o-/p- Chlorobenzoic acids and o-/p- Nitroanilines. Separation Science and Technology, 39: 439-459.