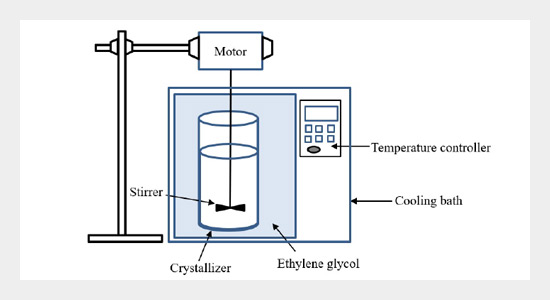
REFERENCES
- [1] Othman, N. H., Alias, N. H., Shahruddin, M. Z., Abu Bakar, N. F., Nik Him, N. R. and Lau, W. J. 2018. Adsorption kinetics of methylene blue dyes onto magnetic graphene oxide. Journal of Environmental Chemical Engineering, 6:2803-2811.
- [2] Kariyajjanavar, P., Narayana, J. and Nayaka, Y. A. 2013. Degradation of textile dye C.I. Vat Black 27 by electrochemical method by using carbon electrodes. Journal of Environmental Chemical Engineering, 1:975-980.
- [3] He, J., Cui, A., Deng, S. and Chen, J. P. 2018. Treatment of methylene blue containing wastewater by a cost-effective micro-scale biochar/polysulfone mixed matrix hollow fiber membrane: Performance and mechanism studies. Journal of Colloid and Interface Science, 512:190-197.
- [4] Novais, R. M., Ascensão, G., Tobaldi, D. M., Seabra, M. P. and Labrincha, J. A. 2018. Biomass fly ash geopolymer monoliths for effective methylene blue removal from wastewaters. Journal of Cleaner Production, 171:783-794.
- [5] Fadzil, F., Ibrahim, S. and Hanafiah, M. A. K. M. 2016. Adsorption of lead(II) onto organic acid modified rubber leaf powder: Batch and column studies. Process Safety and Environmental Protection, 100:1-8.
- [6] Jeon, C. 2017. Adsorption and recovery of immobilized coffee ground beads for silver ions from industrial wastewater. Journal of Industrial and Engineering Chemistry, 53:261-267.
- [7] Alias, N. H., Jaafar, J., Samitsu, S., Yusof, N., Othman, M. H. D., Rahman, M. A., Ismail, A. F., Aziz, F., Salleh, W. N. W. and Othman, N. H. 2018. Photocatalytic degradation of oilfield produced water using graphitic carbon nitride embedded in electrospun polyacrylonitrile nanofibers. Chemosphere, 204:79-86.
- [8] Gade, R., Ahemed, J., Yanapu, K. L., Abate, S. Y., Tao, Y.-T. and Pola, S. 2018. Photodegradation of organic dyes and industrial wastewater in the presence of layer-type perovskite materials under visible light irradiation. Journal of Environmental Chemical Engineering, 6:4504-4513.
- [9] Petrinic, I., Korenak, J., Povodnik, D. and Hélix-Nielsen, C. 2015. A feasibility study of ultrafiltration/reverse osmosis (UF/RO)-based wastewater treatment and reuse in the metal finishing industry. Journal of Cleaner Production, 101:292-300.
- [10] Ochando-Pulido, J. M. and Martinez-Ferez, A. 2017. Experimental design optimization of reverse osmosis purification of pretreatedolive mill wastewater. Science of The Total Environment, 587-588, 414-422.
- [11] Volpin, F., Fons, E., Chekli, L., Kim, J. E., Jang, A. and Shon, H. K. 2018. Hybrid forward osmosis-reverse osmosis for wastewater reuse and seawater desalination: Understanding the optimal feed solution to minimise fouling. Process Safety and Environmental Protection, 117:523-532.
- [12] Hasan, M., Rotich, N., John, M. and Louhi-Kultanen, M. 2017. Salt recovery from wastewater by air-cooled eutectic freeze crystallization. Chemical Engineering Journal, 326:192-200.
- [13] Jusoh, M., Mohd Yunus, R. and Abu Hassan, M. A. 2008. Effect of Flowrate and Coolant Temperature on the Efficiency of Progressive Freeze Concentration on Simulated Wastewater, World Academy of Science, Engineering and Technology.
- [14] Yin, Y., Yang, Y., de Lourdes Mendoza, M., Zhai, S., Feng, W., Wang, Y., Gu, M., Cai, L. and Zhang, L. 2017. Progressive freezing and suspension crystallization methods for tetrahydrofuran recovery from Grignard reagent wastewater. Journal of Cleaner Production, 144:180-186.
- [15] Lu, H., Wang, J., Wang, T., Wang, N., Bao, Y. and Hao, H.. 2017. Crystallization techniques in wastewater treatment: An overview of applications. Chemosphere, 173:474-484.
- [16] Tung, H., Paul, L., Midler, M. and McCauley, J. A. 2008. Critical Issues in Crystallization Practice, Crystallization of Organic Compounds.
- [17] Osorio, M., Moreno, F. L., Raventós, M., Hernández, E. and Ruiz, Y. 2018. Progressive stirred freeze-concentration of ethanol-water solutions. Journal of Food Engineering, 224:71-79.
- [18] Sánchez, J., Ruiz, Y., Auleda, J. M., Hernández, E. and Raventós, M. 2009. Review. Freeze Concentration in the Fruit Juices Industry. Food Sci Technol Int, 15:303-315.
- [19] Moreno, F. L., Raventós, M., Hernández, E. and Ruiz, Y. 2014. Block freeze-concentration of coffee extract: Effect of freezing and thawing stages on solute recovery and bioactive compounds. J Food Eng, 120:158-166.
- [20] Safiei, N. Z., Ngadi, N., Johari, A., Zakaria, Z. Y. and Jusoh, M. 2017. Grape Juice Concentration by Progressive Freeze Concentrator Sequence System. Journal of Food Processing and Preservation, 41, e12910-n/a.
- [21] Hamid, F. H., Rahim, N. A., Johari, A., Ngadi, N., Zakaria, Z. Y. and Jusoh, M. 2015. Desalination of seawater through progressive freeze concentration using a coil crystallizer. Water Science and Technology: Water Supply, 15:625-631.
- [22] Amran, N. A. and Jusoh, M. 2016. Effect of coolant temperature and circulation flowrate on the performance of a vertical finned crystallizer. Procedia Engineering, 148:1408-1415.
- [23] Orellana-Palma, P., Petzold, G., Pierre, L. and Pensaben, J. M. 2017. Protection of polyphenols in blueberry juice by vacuum-assisted block freeze concentration. Food Chem Toxicol, 109:1093-1102.
- [24] Jaster, H., Arend, G. D., Rezzadori, K., Chaves, V. C., Reginatto, F. H. and Petrus, J. C. C. 2017. Enhancement of antioxidant activity and physicochemical properties of yogurt enriched with concentrated strawberry pulp obtained by block freeze concentration. Food Research International.
- [25] Petzold, G., Orellana, P., Moreno, J., Cerda, E. and Parra, P. 2016. Vacuum-assisted block freeze concentration applied to wine. Innovative Food Science & Emerging Technologies, 36:330-335.
- [26] Miyawaki, O., Omote, C., Gunathilake, M., Ishisaki, K., Miwa, S., Tagami, A. and Kitano, S. 2016. Integrated system of progressive freeze-concentration combined with partial ice-melting for yield improvement. J Food Eng, 184:38-43.
- [27] Miyawaki, O., Gunathilake, M., Omote, C., Koyanagi, T., Sasaki, T., Take, H., Matsuda, A., Ishisaki, K., Miwa, S. and Kitano, S. 2016. Progressive freeze-concentration of apple juice and its application to produce a new type apple wine. Journal of Food Engineering, 171: 153-158.
- [28] Gunathilake, M., Dozen, M., Shimmura, K. and Miyawaki, O. 2014. An apparatus for partial ice-melting to improve yield in progressive freeze-concentration. Journal of Food Engineering, 142:64-69.
- [29] Shirai, Y., Wakisaka, M., Miyawaki, O. and Sakashita, S. 1998. Conditions of producing an ice layer with high purity for freeze wastewater treatment. J Food Eng, 38:297-308.
- [30] Lorain, O., Thiebaud, P., Badorc, E. and Aurelle, Y. 2001. Potential of freezing in wastewater treatment: soluble pollutant applications. Water Res, 35:541-547.
- [31] Miyawaki, O., Liu, L., Shirai, Y., Sakashita, S. and Kagitani, K. 2005. Tubular ice system for scale-up of progressive freeze-concentration. J Food Eng, 69:107-113.
- [32] Jusoh, M., Mohd Yunus, R. and Abu Hassan, M. A. 2009. Performance Investigation on a New Design for Progressive Freeze Concentration System. Journal of Applied Sciences, 9:3171-3175.
- [33] Samsuri, S., Amran, N. A. and Jusoh, M. 2015. Spiral finned crystallizer for progressive freeze concentration process. Chem Eng Res Des, 104:280-286.
- [34] Luo, C. S., Chen, W. W. and Han, W. F. 2010. Experimental study on factors affecting the quality of ice crystal during the freezing concentration for the brackish water. Desalination, 260:231-238.
- [35] Garg, V. K., Amita, M., Kumar, R. and Gupta, R. 2004. Basic dye (methylene blue) removal from simulated wastewater by adsorption using Indian Rosewood sawdust: a timber industry waste. Dyes and Pigments, 63:243-250.
- [36] Melak, F., Du Laing, G., Ambelu, A. and Alemayehu, E. 2016. Application of freeze desalination for chromium (VI) removal from water. Desalination, 377:23-27.
- [37] Liu, L., Fuji, T., Hayakawa, K. and Miyawaki, O. 1998. Prevention of Initial Supercooling in Progressive Freeze Concentration. Bioscience, Biotechnology and Biochemistry, 62:2467-2469.
- [38] Zhang, Z. and Hartel, R. W. 1996. A multilayer freezer for freeze concentration of liquid milk. J Food Eng, 29:23-38.
- [39] Yang, Y., Lu, Y., Guo, J. and Zhang, X. 2017. Application of freeze concentration for fluoride removal from water solution. Journal of Water Process Engineering, 19:260-266.
- [40] Fukui, K. and Maeda, K. 2002. Effects of crystal growth rate and heat and mass transfer on solute distribution. Chemical Engineering Science, 57:3133-3140.
- [41] Fujioka, R., Wang, L. P., Dodbiba, G. and Fujita, T. 2013. Application of progressive freeze-concentration for desalination. Desalination, 319:33-37.
- [42] Sánchez, J., Ruiz, Y., Raventós, M., Auleda, J. M. and Hernández, E. 2010. Progressive freeze concentration of orange juice in a pilot plant falling film. Innovative Food Science & Emerging Technologies, 11:644-651.
- [43] Zambrano, A., Ruiz, Y., Hernández, E., Raventós, M. and Moreno, F. L. 2018. Freeze desalination by the integration of falling film and block freeze-concentration techniques. Desalination, 436:56-62.