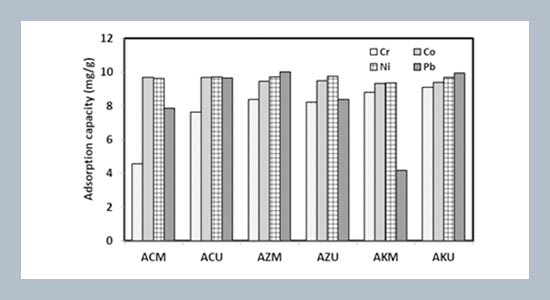
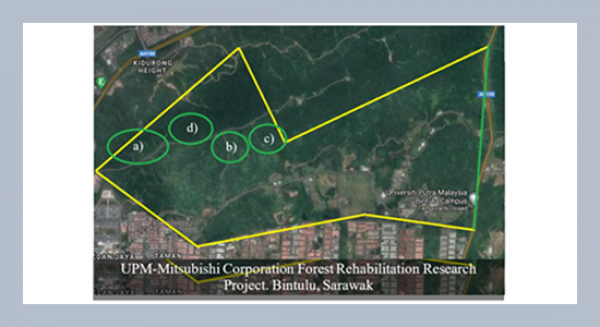
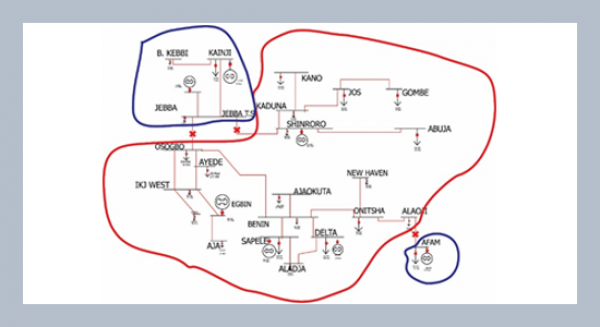
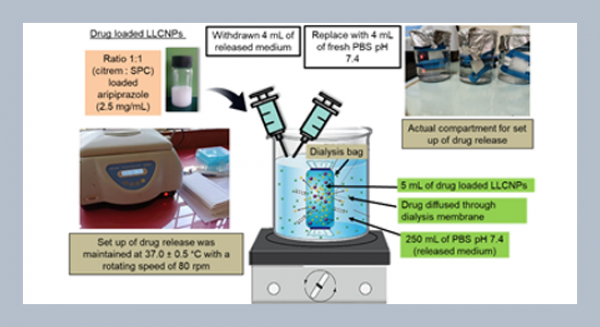
REFERENCES
- Akkaya, R., Ulusoy, U. 2011. Adsorption of lead on to poly (acrylamide-maleic acid)-based hydrogel composites, Hacettepe Journal of Biology and Chemistry, 39, 257–264.
- Al-Abachi, M.Q. 2013. Evaluation of poly acrylic acid (PAA) hydrogel beads as adsorbent for the removal of lead(II)ion from water, Journal of Al-Nahrain University Science, 16, 30–39.
- Atia, A.A., Donia, A.M., Hussin, R.A., Rashad, R.T. 2009. Swelling and metal ion uptake characteristics of kaolinite containing poly [(acrylic acid)-co-acrylamide] hydrogels, Desalination and Water Treatment, 3, 73–82.
- Ayawei, N., Ebelegi, A.N., Wankasi, D. 2017. Modelling and interpretation of adsorption isotherms, Journal of Chemistry, 2017, 1–11.
- Barakat, M.A. 2011. New trends in removing heavy metals from industrial wastewater, Arabian Journal of Chemistry, 4, 361–377.
- Bhatia, M., Rajulapati, S.B., Sonawane, S., Girdhar, A. 2017. Synthesis and implication of novel poly (acrylic acid)/nanosorbent embedded hydrogel composite for lead ion removal, Scientific Reports, 7, 1–16.
- Bulut, Y., Akçay, G., Elma, D., Serhatli, I.E. 2009. Synthesis of clay-based superabsorbent composite and its sorption capability, Journal of Hazardous Materials, 171, 717–723.
- Chen, F., Ye, W., Tang, Y. 2012. Adsorption of heavy metals by sodium polyacrylate-humic acid-rectorite composite as a novel adsorbent, Advanced Materials Research, 550–553, 2428–2435.
- Dean, J.G., Bosqui, F.L., Lanouette, K.H. 1972. Removing heavy metals from waste water, Environmental Science and Technology, 6, 518–522.
- Djelad, A., Morsli, A., Robitzer, M., Bengueddach, A., Di Renzo, F., Quignard, F. 2016. Sorption of Cu(II) ions on chitosan-zeolite X composites: Impact of gelling and drying conditions, Molecules, 21, 1–15.
- El-Kamash, A.M., Zaki, A.A., El Geleel, M.A. 2005. Modeling batch kinetics and thermodynamics of zinc and cadmium ions removal from waste solutions using synthetic zeolite A, Journal of Hazardous Materials, 127, 211–220.
- Elsayed, M.M. 2019. Hydrogel preparation technologies: Relevance kinetics, thermodynamics and scaling up aspects, Journal of Polymers and the Environment, 27, 871–891.
- Emamjomeh, M.M., Sivakumar, M. 2009. Review of pollutants removed by electrocoagulation and electrocoagulation/flotation processes, Journal of Environmental Management, 90, 1663–1679.
- Esalah, J.O., Weber, M.E., Vera, J.H. 1999. Removal of lead from aqueous solutions by precipitation with sodium di-(n-octyl) phosphinate, Separation and Purification Technology, 18, 25–36.
- Foo, K.Y., Hameed, B.H. 2010. Insights into the modeling of adsorption isotherm systems, Chemical Engineering Journal, 156, 2–10.
- Ghurye, G., Clifford, D., Tripp, A. 2004. Iron coagulation and direct microfiltration to remove arsenic from groundwater, American Water Works Association, 96, 143–152.
- Gomez-Maldonado, D., Erramuspe, I.B.V., Peresin, M.S. 2019. Natural polymers as alternative adsorbents and treatment agents for water remediation, BioResources, 14, 10093–10160.
- Green, D.W. 2008. Perry’s chemical engineers’ handbook, McGraw-Hill, New York.
- Hani, H.A. 2010. Studies on zeolites for heavy metals removal, PhD. Thesis, Faculty of Engineering, Cairo Univeristy, Egypt. http://www.eulc.edu.eg/eulc_v5/libraries/Start.aspx?ScopeID=1.&fn=ApplySearch&SearchIdForm=&ScopeIDSelect=1.&ItemType=24.2.5.&SearchText1=Heba+hani&criteria1=1.&opr1=and&SearchText2=Zeolite&criteria2=1.&opr2=and&SearchText3=Heavy+metals&criteria3=1.&SearchTextFT=&Generation_Term=&BibID=&PublishYear=&OrderKey=&PageSize=10
- He, Y., Pei, M., Xue, N., Wang, L., Guo, W. 2016a. Synthesis of sodium polyacrylate-bentonite using: In situ polymerization for Pb2+ removal from aqueous solutions, RSC Advances, 6, 48145–48154.
- He, S., Zhang, F., Cheng, S., Wang, W. 2016b. Synthesis of sodium acrylate and acrylamide copolymer/GO hydrogels and their effective adsorption for Pb2+ and Cd2+, ACS Sustainable Chemistry and Engineering, 4, 3948–3959.
- Inagaki, S., Yokoi, T., Kubota, Y., Tatsumi, T. 2007. Unique adsorption properties of organic-inorganic hybrid zeolite IEZ-1 with dimethylsilylene moieties, Chemical Communications, 1, 5188–5190.
- Liu, M., Li, W., Rong, J., Zhou, C. 2012. Novel polymer nanocomposite hydrogel with natural clay nanotubes, Colloid and Polymer Science, 290, 895–905.
- Liu, Q., Yang, B., Zhang, L., Huang, R. 2015. Adsorptive removal of Cr(VI) from aqueous solutions by cross-linked chitosan/bentonite composite, Korean Journal of Chemical Engineering, 32, 1314–1322.
- Mahmoud, A., Hoadley, A.F.A. 2012. An evaluation of a hybrid ion exchange electrodialysis process in the recovery of heavy metals from simulated dilute industrial wastewater, Water Research, 46, 3364–3376.
- Natkański, P., Białas, A., Kuśtrowski, P. 2012. The synthesis of poly (acrylic acid)-bentonite: And polyacrylamide-bentonite composites: For adsorption applications, Chemik, 66, 742–749.
- Nechifor, G., Pascu, D.E., Neagu, M.P., Traistaru, G.A., Albu, P.C. 2015. Comparative study of temkin and floryhuggins isotherms for adsorption of phosphate anion on membranes, UPB Scientific Bulletin, Series B: Chemistry and Materials Science, 77, 63–72.
- Qiu, H., Lv, L., Pan, B.C., Zhang, Q.J., Zhang, W.M., Zhang, Q.X. 2009. Critical review in adsorption kinetic models, Journal of Zhejiang University: Science A, 10, 716–724.
- Rafiei, H.R., Shirvani, M., Ogunseitan, O.A. 2016. Removal of lead from aqueous solutions by a poly (acrylic acid)/bentonite nanocomposite, Applied Water Science, 6, 331–338.
- Renu, M.A., Singh, K. 2017. Heavy metal removal from wastewater using various adsorbents: A review, Journal of Water Reuse and Desalination, 7, 387–419.
- Sezgin, N., Balkaya, N. 2016. Adsorption of heavy metals from industrial wastewater by using polyacrylic acid hydrogel, Desalination and Water Treatment, 57, 2466–2480.
- Shikuku, V.O., Kowenje, C.O., Kengara, F.O. 2018. Errors in parameters estimation using linearized adsorption isotherms: Sulfadimethoxine adsorption onto kaolinite clay, Chemical Science International Journal, 23, 1–6.
- Sorour, M.H., Shaalan, H.F., Hani, H.A., Sayed, E.S. 2018. Batch and dynamic investigations on magnesium separation by ion exchange adsorption: Performance and cost evaluation. 20th International Conference on Energy, Environmental and Chemical Engineering, Dubai, 1702–1711.
- Sorour, M.H., Tewfik, S.R., Abulnour, A.G., Shaalan, H.F., Hani, H.A., Al‐Bazedi, G.A. 2020. Development and assessment of adsorptive hydrogel: Process engineering and economic considerations, Engineering Reports, 2, 1–13.
- Wan Ngah, W.S., Teong, L.C., Hanafiah, M.A.K.M. 2011. Adsorption of dyes and heavy metal ions by chitosan composites: A review, Carbohydrate Polymers, 83, 1446–1456.
- Wang, J., Guo, X. 2020. Adsorption kinetic models: Physical meanings, applications, and solving methods, Journal of Hazardous Materials, 390, 122156.
- Yakout, S.M., Elsherif, E. 2010. Batch kinetics, isotherm and thermodynamic studies of adsorption of strontium from aqueous solutions onto low cost rice-straw based carbons, Carbon - Science and Technology, 3, 144–153.
- Yu, Y., Peng, R., Yang, C., Tang, Y. 2015. Eco-friendly and cost-effective superabsorbent sodium polyacrylate composites for environmental remediation, Journal of Materials Science, 50, 5799–5808.
- Yurlova, L., Kryvoruchko, A., Kornilovich, B. 2002. Removal of Ni(II) ions from wastewater by micellar-enhanced ultrafiltration, Desalination, 144, 255–260.
- Zhang, J., Wang, A. 2015. Polysaccharide-based composite hydrogels for removal of pollutants from water. Advanced separations by specialized sorbents, Chromatographic series, Boca Raton, CRC Press/Taylor & Francis Group.
- Zhao, G., Zhang, H., Fan, Q., Ren, X., Li, J., Chen, Y., Wang, X. 2010. Sorption of copper(II) onto super-adsorbent of bentonite-polyacrylamide composites, Journal of Hazardous Materials, 173, 661–668.