REFERENCES
- [1] Narin, I., Surme, Y., Soylak, M., and Dogan, M. 2006. Speciation of Cr(III)and Cr(VI) in environmental samples by solid phase extraction on Ambersorb 563 resin. Journal of Hazardous Materials, B136: 579-584.
- [2] Banks, M. K., Schwab, A. P., and Henderson, C. 2006. Leaching and reduction of chromium in soil as affected by soil organic content and plants. Chemosphere, 62: 255-264.
- [3] Narin, I., Kars, A., and Soylak, M. 2008. A novel solid phase extraction procedure on Amberlite XAD-1180 for speciation of Cr(III), Cr(VI) and total chromium in environmental and pharmaceutical samples. Journal of Hazardous Materials, 150: 453-458.
- [4] Puzon, G. J., Tokala, R. K., Zhang, H., Yonge, D., Peyton, B. M., and Xun, L. 2008. Mobility and recalcitrance of organo-chromium ((III) complexes. Chemosphere, 70: 2054-2059.
- [5] Uluozlu, O. D., Tuzen, M., Mendil, D., Kahveci, B., and Soylak, M. 2009. 3-Ethyl-4-(p-chlorobenzylidenamino-4,5-dihydro1H-1,2,4-triazol-5-one(EPHBAT) as preci -pitant for carrier element free coprecipitation and speciation of chromium(III) and chromium(VI). Journal of Hazardous Materials, 172: 395-399.
- [6] Isoyama, M. and Wada, S. I. 2006. Soil Chemistry Effect on Feasibility of Cr-decontamination by Acid-Washing. J. Fac. Agr., Kyushu Univ., 51: 33-36.
- [7] Evanko, C. R. and Dzombak, D. A. 1997. Remediation of metals-contaminated soils and groundwater. Technology Evaluation Report, TE-97-01, Ground-water Remediation Technologies Analysis Center, USA.
- [8] Laborda, F., Gorriz, M. P., Bolea, E., and Castillo, J. R. 2007. Mobilization and speciation of chromium in compact: a methodological approach. Science of The Total Environment, 373: 383-390.
- [9] Dermont, G., Bergeron, M., Mercier, G., and Richer-Lafleche, M. 2008. Soil washing for metal removal: A review of physical/chemical technologies and field applications. Journal of Hazardous Materials, 152: 1-31.
- [10] Kuo, S., Lai, M. S., and Lin, C. W. 2006. Influence of solution acidity and CaCl2 concentration on the removal of heavy metals from metal-contaminated rice soils. Environmental Pollution, 144: 918-925.
- [11] Abumaizar, R. J. and Smith, E. H. 1999. Heavy metal contaminants removal by soil washing. Journal of Hazardous Material, B70: 71-86.
- [12] Neale, C. N., Bricka, R. M., and Chao, A. C. 1997. Evaluating acid and chelating agents for removing heavy metals from contaminated soils. Environmental Progress, 16: 274-280.
- [13] Jean, L., Bordas, F., and Bollinger, J. C. 2007. Chromium and nickel mobilization from a contaminated soil using chelants. Environmental Pollution, 147: 729-736.
- [14] Kantar, C. and Honeyman, B. D. 2006. Citric acid enhanced remediation of soils contaminated with uranium by soil flushing and soil washing. Journal of Environmental Engineering, 132: 247-255.
- [15] Lenhart, J. J., Cabaniss, S. E., MacCarthy, P., and Honeyman, B. D. 2000. Uranium (VI) complexation with citric, humic and fulvic acids. Acta, 86: 345-353.
- [16] Peters, R. W. 1999. Chelant extraction of heavy metals from contaminated soils. Journal of Hazardous Material, 66: 151-210.
- [17] Logus, B. A., Smith, R. W., and Westall, J. C. 2004. Role of surface alteration in determining the mobility of U(VI) in the presence of citrate: implications for extraction of U(VI) from soils. Sci. Technol., 38: 3752-3759.
- [18] Mason, C. F. V., Turney, W. R. J. R., Thomson, B. M., Lu, N., Longmire, P. A., and Chisholm-Brause, C. J. 1997. Carbonate leaching of uranium from contaminated soils. Environ. Sci. Technol., 31: 2707-2711.
- [19] Francis, C. W., Timpson, M. E., and Wilson, J. H. 1999. Bench-and pilot-scale studies relating to the removal of uranium from uranium-contaminated soils using carbonate and citrate lixiviants. Journal of Hazardous Material, 66: 67-87.
- [20] Li, F. Y. and Cheng, S. X. 2007. “Laboratory Manual for Soil Analysis”. New Wun Ching Developmental Publishing, Taiwan.
- [21] Tessier, A., Campbell P. G. C., and Bisson, M. 1979. Sequential extraction procedure for the speciation of particulate trace metals. Analytical Chemistry, 51: 844-850.
- [22] Nyamangara, J. 1998. Use of sequential extraction to evaluate zinc and copper in a soil amended with sewage sludge and inorganic metal salts. Agriculture Ecosystems and Environment, 69: 135-141.
- [23] Tokalioglu, S., Kartal, S., and Elci, L. 2000. Determination of heavy metals and their speciation in lake sediments by flame atomic absorption spectrometry after a four-stage sequential extraction procedure. Analytica Chimica Acta, 413: 33-40.
- [24] Mossop, K. F. and Davidson, C. M. 2003. Comparison of original and modified BCR sequential extraction procedures for the fractionation of copper, iron, lead, manganese and zinc in soil and sediments. Analytica Chimica Acta, 478: 111-118.
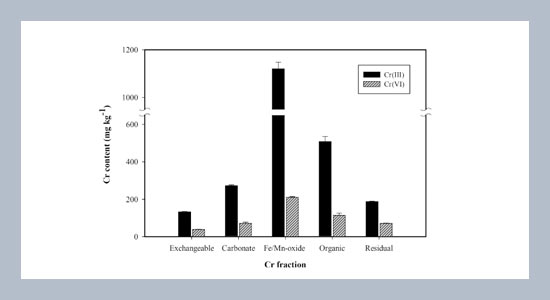
- [25] Koleli, N. 2004. Speciation of chromium in 12 agricultural soils from Turkey. Chemosphere, 57: 1473-1478.
- [26] Weng, H. P. 2004. Leaching treatment of metal contaminated farm soil and investigation the characteristic change of the soil. Ms.D. diss., National Chung Hsing University, Taiwan.
- [27] Fu, W. C. 2006. A study of the distribution and the transformation of metal speciation in contaminated agro-environment exemplified by Cr, Cu, Zn, Ni, Pb, and Cd. Ms.D. diss., National Chung Hsing University, Taiwan.
- [28] Eary, L. E. and Rai, D. 1988. Chromate removal from aqueous wastes by reduction with ferrous ion. Sci. Technol., 22: 972-977.
- [29] Manceau, A., Schlegel, M. L., Musso, M., Sole, V. A., Gauthier, C., Pettt, P. E., and Trolard, F. 2000. Crystal chemistry of trace elements in natural and synthetic goethite. Geochimica Cosmochimica Acta, 64: 3643-3661.
- [30] James, B. R., Petura, J. C., and Vitale, R. J. 1995. Mussoline GR. Hexavalent chromium extraction from soils: a comparison of five methods. Environ. Sci. Technol., 29: 2377-2381.