Sirinapa Arenamnarta and Wimonrat Trakarnprukb* a Program of Petrochemistry and Polymer Science, Faculty of Science, Chulalongkorn University, Bangkok, Thailand
b Functional Polymer and Petrochemistry Research Unit, Department of Chemistry, Faculty of Science, Chulalongkorn University, Bangkok, Thailand
Download Citation:
|
Download PDF
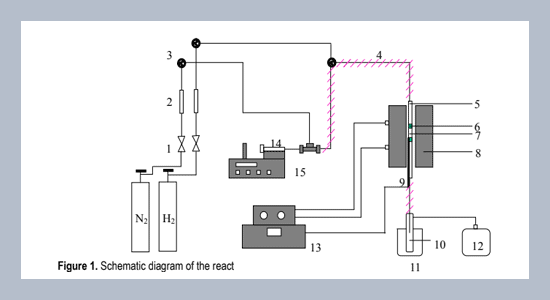
The aqueous ethanol conversion was investigated on dealuminated mordenite (DM) and a series of DM/metal catalysts prepared by impregnation (IMP) and solid-state ion exchange (SSIE). The catalysts were characterized by X-ray fluorescence (XRF), X-ray diffraction (XRD), Fourier transform infrared spectroscopy (FT-IR), nitrogen adsorption method (BET). The conversion of aqueous ethanol (10.0%) on mordenite/metal catalysts was compared at temperature of 350°C. reaction time of 1 h, WHSV (weight hourly space velocity) of 1 h-1 and catalyst weight of 1 g. The product is mainly ethylene with small quantity of other light olefins. At higher temperature (550°C) methane was formed as a result of decomposition reaction. Among several metals (Zn, Mn, Co, Rh, Ni, Fe and Ag) which were incorporated onto the mordenite to prepare DM/single and mixed metal catalysts tested in this work, it was found that DM/Zn and DM/Zn-Ag catalysts prepared by impregnation method gave the highest selectivity to ethylene. For DM/Ni catalyst, it showed high selectivity to ethane due to its hydrogenation ability.ABSTRACT
Keywords:
ethanol conversion; metal-supported mordenite; catalyst; ethylene
Share this article with your colleagues
[1] Tsao, U. and Reilly J. W. 1978. Dehydrate Ethanol to Ethylene. Hydrocarbon Processing, 133-136.REFERENCES
[2] Le Van Mao, R., Nguyen, T. M., and McLaughlin, G. P. 1989. The bioethanol-to-ethylene (B.E.T.E.) process. Applied Catalysis, 48, 265-277.
[3] Aldridge, A. Al Veryklos, X. E., and Mutharasan, R. 1984. Recovery of ethanol from fermentation broths by catalytic conversion to gasoline. 2. Energy analysis. Industrial & Engineering Chemistry Process Design Dev. 23, 733-737.
[4] Bun, S., Nishiyama, S., Tsuruya, S., and Masai, M. 1990. Ethanol Conversion over ion-exchanged ZSM-5 zeolites. Applied Catalysis A. 59, 13-29.
[5] Nguyen, T. M. and Van Mao, R. Le. 1990. Conversion of ethanol in aqueous solution over ZSM-5 zeolite study of the reaction network. Applied Catalysis A. 58, 119-129.
[6] Sawa, M., Kato, K., Hirota, K., Niwa, M., and Murakami, Y. 1990. Mordenite with long life and selectivity for methanol conversion to gasoline: mordenite modified by barium ion exchange, dealumination and chemical vapor deposition of silicon methoxide. Applied Catalysis A. 64, 297-308.
[7] Canizares, P., Lucas, A., Dorado, F., Duran, A., and Asencio, 1998. Characterization of Ni and Pd supported on H-mordenite catalysts: Influence of the metal loading method. Applied Catalysis A. 169, 137-150.
[8] Cartlidge, Sl and Patel, R.1989. in Jacobs, P.A. and van Santen, R.A. (Editors), “Zeolites: Facts, Figures, Future. Stud. Surface Science Catalysis”. 49B, Elsevier, Amsterdam, 1151.
[9] Drozdova, L; Prins, R. 2002. Bonding of Co Ions in ZSM-5, Ferrierite, and Mordenite: An X-ray Absorption, UV-Vis, and IR Study. Journal of Physical Chemistry B, 106, 2240-2248.
[10] Canizares, P., Lucas, A., Dorado, F.,and Asencio, D. 1998. Characterization of Ni and Pd supported on H-mordenite catalysts: Influence of the metal loading method. Applied Catalysis A. 169, 137-150.
[11] Kinger, G., Lugstein, A. and Swagera, R. 2000. Comparison of impregnation, liquid- and solid-state ion exchange procedures for the incorporation of nickel in HMFI, HMOR and HBEA. Activity and selectivity in n-nonane Hydroconversion. Microporous Mesoporous Materials, 39, 307-317.
[12] Juguin, B. Hughes, F., and Hamon, C. 1992. Selective production of Light Olefins by Zeolite-Catalyzed Methanol Conversion. in: Albright, L.F., Crynes, B.L.,and Nawak, S. (Eds.), “Novel production methods for ethylene light hydrocarbons and aromatics, Chemistry Industry”. 46, Dekker, New York, 381-390.
[13] Tabak, S. and Yurchak, S.1990. Conversion of methanol over ZSM-5 to fuels and chemicals. Catalysis Today 6, 307-327.
[14] Sawa, M., Niwa, M., and Murakami, Y. 1989. Acid-leached dealuminated mordenite: Effect of acid concentration on catalyst life in methanol conversion. Applied Catalysis A. 53, 169-181.
[15] Choo, H., Hong, S., and Kevan, L. 2001. Comparative ESR and catalytic studies of ethylene dimerization on Pd(II) exchanged Clinoptilolite, Mordenite, Ferrierite, and SUZ-4. Journal of Physical Chemistry B, 105, 7730-7738.
[16] Marchi, A. and Froment, G. 1993. Catalytic conversion of methanol into light alkanes on Mordenite-like zeolites. Applied Catalysis A. 94, 91-106.
[17] Iomata, M., Yamada, M., Okada, S., Niwa, M., and Murakami, Y. 1986. Benzene-filled pore method: A method of measuring external surface areas applicable to zeolites with low-to-high Si-to-Al ratios. Journal of Catalysis, 100, 264-269.
[18] Breck, D. W. 1974. “Zeolite molecular sieves: structure chemistry and use”. New York: John Wiley & Sons.
[19] Ostroumov, M. and Corona-Chavez, P. 2003. Mineralogical study of mordenite from the Sierra Madre del Sur, Southwestern Mexico. Revista Mexicana de Ciencias Geologicas, 20, 133-138.
[20] Majdan, M., Kawalska-Ternes, M., and Pikus, S. 2003. Vibrational and scanning electron microscopy study of the mordenite modified by Mn, Co, NI, Cu, Zn and Cd. Journal of Molecular Structure. 649, 279-285.
[21] Breen, J. P., Burch, R., and Coleman, H. M. 2002. Metal-catalysed steam reforming of ethanol in the production of hydrogen for fuel cell applications. Applied Catalysis B-Environmental, 39, 65-74.
[22] Mohamed, M. Catalytic properties of Fe ion-exchanged Mordenite toward the ethanol transformation: Influence of the methods of preparation. Journal of Molecular Catalysis A. 2003, 200, 301-313.
[23] Inui, T. and Kang, M. 1997. Reliable procedure for the synthesis of Ni-SAPO-34 as a highly selective catalyst for methanol to ethylene conversion. Applied Catalysis A. 64, 211-223.
[24] Mao, R., Levesque, P., McLaughlin, G., and Dao, L. Ethylene from ethanol over zeolite catalysts. Applied Catalysis A. 34, 163-179.
[25] Choudbury, S., Halik, C., and Lercher, J. 1990. Reactions of ethanol over HZSM-5 Journal of Molecular Catalysis 1990, 62, 289-295.
[26] Oudejans, J. C., Van Den Oosterkap, P. F., and Van Bekkum, H. 1982. Conversion of ethanol over zeolite H-ZSM-5 in the presence of water. Applied Catalysis A. 1982, 3, 109-115.
[27] Kang, M. and T. Inui. 1999. Synthesis of NiAPSO-34 catalysts containing a larger concentration of Ni and effect of its sulfidation on methanol conversion. Journal of Molecular Catalysis, 144, 329-335.
[28] Chang, C.D., C.T-W. Chu, and R.F. Socha. 1984. Methanol conversion to olefins over ZSM-5: I. Effect of temperature and zeolite SiO2/Al2O Journal of Catalysis, 86, 289-296.
[29] Aguayo, A. T., Gayabo, A. G., Tarrio, A. M., and Atutxa, A. 2002. Study of operating variables in the transformation of aqueous ethanol into hydrocarbons on an HZSM-5 zeolite. Journal of Chemical Technology and Biotechnology, 77, 211-216.
[30] Hagen, J. 1999. Industrial catalysis. Germany: WILEY-VCH, 229.
[31] Fougerit, J., Gnep, N., and Guisnet, M. 1990. Selective transformation of methanol into light olefins over a mordenite catalyst: Reaction scheme and mechanism. Microporous and Mesoporous Materials, 29, 79-89.
[32] Gayubo, A., Tarrio, A., Aguayo, A., Olazar, M., and Bilbao, J. 2001. Kinetic modelling of the transformation of aqueous ethanol into hydrocarbons on a HZSM-5 zeolite. Industrial & Engineering Chemistry Research, 40, 3467-3474.
ARTICLE INFORMATION
Accepted:
2005-10-28
Available Online:
2006-04-04
Arenamnart. S., Trakarnpruk, W. 2006. Ethanol conversion to ethylene using Metal-Mordenite catalysts. International Journal of Applied Science and Engineering, 4, 21–32. https://doi.org/10.6703/IJASE.2006.4(1).21
Cite this article: